Top 4 Challenges in Pharmaceutical Product Security in Malaysia & Singapore (and How SmartKood Can Help)
Published on 5 March 2025

(Image by Hazelwood from Pexels)
Traceability in pharmaceuticals strengthens drug safety by enabling real-time tracking of products across the supply chain. It also ensures that only safe and approved medicines reach patients, reducing health risks and financial losses.
Introduction
Combating Counterfeit Medicines: Key Challenges in Pharmaceutical Traceability & Authentication in Malaysia & Singapore
Did you know? According to WHO (2024), at least 10% of medicines are either substandard or falsified in low-middle-income countries. Pharmaceutical product traceability and authentication is critical in ensuring patient safety, regulatory compliance, and supply chain efficiency.
In Malaysia and Singapore, pharmaceutical companies face increasing pressure to comply with NPRA (National Pharmaceutical Regulatory Agency) and HSA (Health Sciences Authority) regulations while tackling the rise of counterfeit medicines, high digital implementation costs and fragmented supply chains. In this blog, we explore the top four challenges in pharmaceutical security in Malaysia and Singapore—and how SmartKood helps overcome them.
1. Complex & Evolving Compliance Regulations
Navigating Regulatory Complexity: Overcoming Compliance Challenges in Malaysia & Singapore’s Pharmaceutical Industry

NPRA & HAS as the main governing body in the pharmaceutical industries in Malaysia and Singapore (Images 1 & 2 retrieved from Google Maps, 2024)
Pharmaceutical regulations in Malaysia and Singapore are constantly evolving, requiring companies to stay updated and ensure compliance with stringent laws. In Malaysia, the NPRA mandates Good Distribution Practice THIRD EDITION, 2018 compliance, requiring quality end-to-end traceability for pharmaceuticals. All the while the planning of full serialization adoption is ongoing, and companies must prepare for upcoming regulatory changes. Meanwhile, Singapore’s HSA enforces strict pharmaceutical import and distribution regulations, requiring companies to closely follow their import and distribution regulations involving licenses, GDP compliance, product registrations, quality management systems, accurate documentation, etc.
While nations strive to ensure the safety and integrity of pharmaceutical products for their citizens, the lack of a standardized regulatory framework further complicates matters for pharmaceutical businesses operating regionally. Varying serialization and reporting requirements create challenges for cross-border trade, increasing compliance risks and operational inefficiencies.
SmartKood’s regulatory tracking system keeps pharmaceutical companies updated on NPRA, HSA, and global compliance standards. It’s cloud-based serialization and reporting tools ensure compliance with GDP and international standards such as GS1 and EPCIS. Additionally, its automated audit-ready documentation helps businesses minimize compliance risks and regulatory fines, ensuring smooth operations and providing pharma grade products for their customers.
2. Counterfeit Drugs
Counterfeit Medicines in Malaysia & Singapore: Health Risks, Consumer Trust, and the Need for Stronger Traceability

SmartKood for No More Counterfeit Drugs: Because real medicine saves lives. SmartKood for No More Counterfeit Drugs. (Image by Hazelwood from Pexels)
The presence of counterfeit medicines in the market poses serious health risks to consumers and damages the reputation of legitimate pharmaceutical brands. Numerous reports in 2023 by the Health Sciences Authority (2024) state that severe health conditions were developed after consuming counterfeit medicine that were altered with steroids. Furthermore, according to TheStar (2023), during the first half of 2023, Malaysian law enforcement seized RM12,763,052 worth of counterfeit medicines. Despite strict enforcement, perpetrators still find ways to create loopholes for non-compliant drugs to enter the market, further complicating traceability efforts.
Howarth (2024) has stated that 81% of consumers require trust in a brand to consider making purchases. Counterfeit drugs not only endanger patients but also weaken consumer trust in pharmaceutical brands. Without robust traceability solutions, it is difficult to verify the authenticity of medicines, making it easier for counterfeit products to infiltrate the supply chain and plague the market.
SmartKood addresses this issue by implementing an advanced track-and-trace system, ensuring pinpoint accurate verification of drug authenticity through one swift scan of the QR code. Its smart serialization technology utilises a Dynamic QR system, allowing consumers and regulators to verify product authenticity with ease. Additionally, its state-of-the-art counterfeit detection system identifies suspicious transactions and supply chain anomalies, providing an added layer of protection from price dumping and illicit trades. While SmartKood helps businesses detect unusual activities within the supply chain, consumers can also verify the end-product authenticity by scanning SmartKood’s QR, enhancing customer trust and strengthening brand reputation.
3. Supply Chain Fragmentation & Visibility Issues
Pharmaceutical Supply Chain Challenges in Malaysia & Singapore: Traceability, Cold Chain Logistics, and Compliance Risks
SmartKood for Trust in Every Dose: No fakes, no risks. SmartKood for Trust in Every Dose. (Images by: Goncharenok from Pexels)
Most pharmaceutical supply chains in Malaysia and Singapore often involve multiple stakeholders, including manufacturers, distributors, and pharmacies. This complexity often results in fragmented data silos, making it challenging to achieve real-time visibility. As reported by Dervaux (2024), 69% of companies experience experiences poor visibility across their supply chains. Without proper traceability systems, companies struggle to monitor product movement, leading to inefficiencies, delivery delays, and compliance failures.
Bhusal, Bhandari, Kumari, and Sah (2022) had stated wastage during transportation, expiration, storage errors or contamination after opening vial as the major reasons for vaccine wastages. Pharmaceutical logistics, specifically cold chain logistics further complicate the situation, as most vaccines and biologics require strict temperature control. A lack of real-time tracking and monitoring can compromise the integrity of temperature-sensitive pharmaceuticals, resulting in significant financial losses and potential health risks.
SmartKood’s modern supply chain visibility system allows point-to-point (P2P) monitoring and optimization, reducing inefficiencies and improving operational transparency. By integrating a cloud-based solution, SmartKood enables end-to-end pharmaceutical traceability, enhancing supply chain efficiency. With SmartKood, pharmaceutical companies gain clear supply chain visibility, enabling better logistics and cold supply chain planning while minimizing financial and health risks.
4. High Implementation Costs for SMEs
Pharmaceutical Supply Chain Challenges in Malaysia & Singapore: Traceability, Cold Chain Logistics, and Compliance Risks

Digital transformation in pharmaceuticals is costly but enhances efficiency, reduces risks, and strengthens supply chain integrity. (Image by Miroshnichenko from Pexels)
The cost of implementing traceability solutions can be a major barrier, particularly for small and medium-sized enterprises (SMEs). As reported by the World Bank (2022), one of the main challenges faced by Malaysian SMEs while trying to digitalise is financial constraints. Aside from that, TEKsystems (2024) conducted a survey (see figure 1.0) where 26% of respondents considered high costs as one of the barriers to digital transformations for their businesses. Many pharmaceutical SMEs struggle to adopt serialization technology due to high upfront costs and the lack of in-house expertise to manage digital transformation. As a result, manual processes remain prevalent, slowing down compliance efforts and reducing operational efficiency.
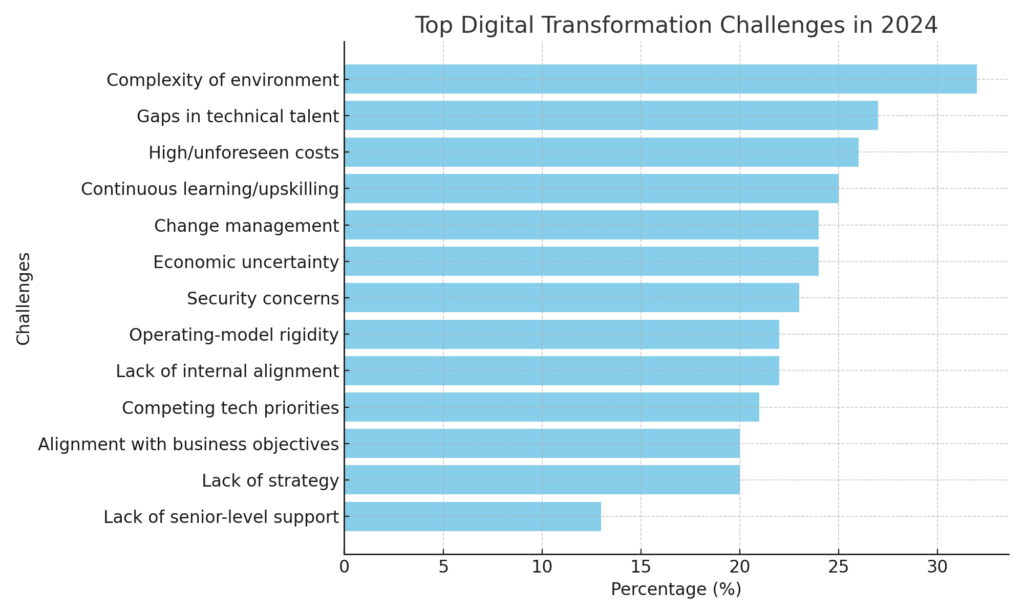
Figure 1.0: Top Digital Transformation Barriers in 2024. (Figure retrieved from TEKsystems, 2024)
SmartKood offers an affordable SaaS-based traceability solution, eliminating the need for significant upfront investments. Its high-tech serialization software is designed to be user-friendly, allowing businesses to implement traceability without requiring extensive IT support. Additionally, SmartKood simplifies regulatory reporting, achieving seamless operations for SMEs looking to scale their traceability efforts. Compared to other traceability solutions that can cost up to $10,000+ in one-time fees, SmartKood offers an affordable digital solution subscription with over 12 primary features for all product traceability needs. From product traceability to price dumping control to anti-counterfeit solutions, SmartKood is the ideal digital solutions for Pharmaceutical SMEs that wants to provide quality healthcare to their customers.
Conclusion
SmartKood for Transparency: Nothing to hide, everything to gain. SmartKood for Transparency.
Ensuring pharmaceutical traceability and authentication is no longer an option—it is a necessity. From evolving regulatory compliance and counterfeit drug risks to supply chain visibility challenges and high digital implementation costs, pharmaceutical companies in Malaysia and Singapore face significant hurdles in safeguarding product integrity.
SmartKood offers an innovative, cost-effective, and comprehensive digital solution to address these challenges. With real-time tracking, smart serialization, and automated compliance tools, SmartKood empowers businesses to strengthen regulatory compliance, protect consumers, and enhance supply chain efficiency. For pharmaceutical companies—especially SMEs—seeking a scalable and affordable track-and-trace solution, SmartKood is the key to ensuring safe, trusted, and high-quality healthcare.
By embracing digital transformation, the industry can take a major step toward eliminating counterfeit medicines, improving supply chain transparency, and securing public health for future generations.
Author: Chung Kai Foong, Team SmartKood.
Looking to enhance your supply chain visibility and product traceability? Ready to transform your supply chain? Learn how SmartKood can help your business stay competitive!
Sources referenced:
Bhusal, A., Bhandari, S., Kumari, N., & Sah, R. P. (2022). COVID vaccine wastage: Double trouble in growing pandemic. https://pmc.ncbi.nlm.nih.gov/articles/PMC9462924
Dervaux, A. (2024). Supply Chain Visibility: Critical Challenges and How ClicData Can Help. https://www.clicdata.com/blog/improving-supply-chain-visibility/
Goncharenok, M. (2021). A close-up shot of COVID-19 vaccine vials on a blue surface [Stock image]. Pexels. https://www.pexels.com/photo/a-close-up-shot-of-covid-19-vaccine-vials-on-a-blue-surface-5995158/
Goncharenok, M. (2021). Silver and blue beaded bracelets [Stock image]. Pexels. https://www.pexels.com/photo/silver-and-blue-beaded-bracelets-5995159/
Google. (2024). Google Maps location [Online map]. Retrieved from https://maps.app.goo.gl/XMTHF28dr5rPXFqH9
Google. (2024). Google Maps location [Online map]. Retrieved from https://maps.app.goo.gl/pGu95f43BDRDu2v38
Hazelwood, S. (2020). Close-up photo of pills [Stock image]. Pexels. https://www.pexels.com/photo/close-up-photo-of-pills-3652103/
Health Sciences Authority. (2024). HSA Seized Over 1.12 Million Units of Illegal Health Products and Removed More Than 12,000 Illegal Product Listings Online in 2023. https://www.hsa.gov.sg/announcements/press-release/hsa-annualsummary2023
Health Sciences Authority. (n.d.). Health Sciences Authority Singapore. https://www.hsa.gov.sg/
Howarth, J. (2024). 33 New Branding Statistics and Trends for 2024. https://explodingtopics.com/blog/branding-stats
Kaboompics.com. (2020). Heap of various pills in blisters on yellow background [Stock image]. Pexels. https://www.pexels.com/photo/heap-of-various-pills-in-blisters-on-yellow-background-4210611/
Miroshnichenko, T. (2021). Banknotes and calculator on table [Stock image]. Pexels. https://www.pexels.com/photo/banknotes-and-calculator-on-table-6694543/
National Pharmaceutical Regulatory Agency. (2018). GUIDELINE ON GOOD DISTRIBUTION PRACTICE. https://npra.gov.my/images/Guidelines_Central/Guidelines_on_Regulatory/2018/GUIDELINE_ON_GDP_3rd_Edi_2018.pdf
TEKsystems. (2024). State of Digital Transformation 2024. https://www.teksystems.com/en/insights/state-of-digital-transformation-2024
TheStar. (2023). Three arrested as cops bust counterfeit medicine lab in Taiping. https://www.thestar.com.my/news/nation/2023/06/16/three-arrested-as-cops-bust-counterfeit-medicine-lab-in-taiping
World Bank. (2022). Digitalizing SMEs to Boost Competitiveness. https://documents1.worldbank.org/curated/en/099515009292224182/pdf/P17608901a9db608909f5b02980d48c4e28.pdf
World Health Organization. (2024). Substandard and falsified medical products. https://www.who.int/news-room/fact-sheets/detail/substandard-and-falsified-medical-products

